- Extract high quality & purity total RNA including miRNA
- No phenol step required - isolate all RNA in one fraction
- Elute extracted total RNA in small volume (20 µL)
- Bind & elute all RNA irrespective of size or GC content, without bias.
- Efficiently extract small RNA irrespective of GC content
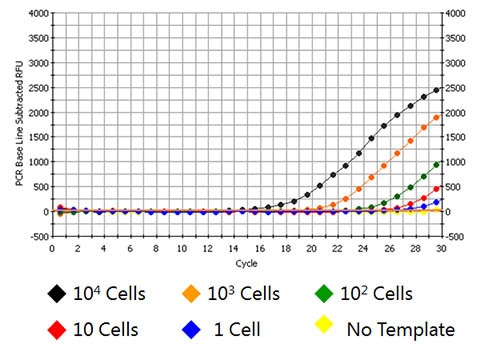
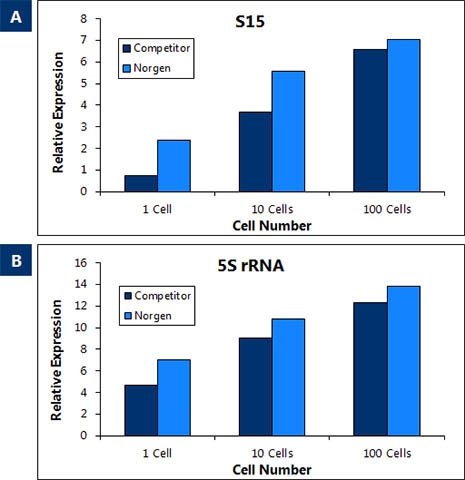
- Very sensitive & linear down to a few cells without the need for carrier RNA
- Convenient & fast spin column format
- Isolate from a wide variety of specimens
- Purified RNA is suitable for a variety of downstream applications, including Small RNA Sequencing. Find out more information on Norgen"s NGS services
- Buffer chemistry inactivates viruses including SARS-CoV-2 - Explore the Application Note
Total RNA Purification Micro Kit
This kit extracts total RNA from low cell number samples and elutes in a convenient 20 µL elutionfor a number of downstream applications.
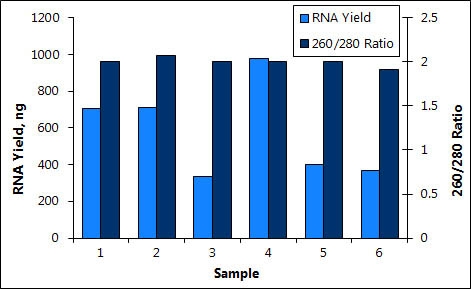
This kit is suitable for the isolation of total RNA from a range of samples including small input of cells, needle biopsies, LCM, CTCand other low cell number samples. Extract high quality and purity RNA with excellent RIN values and A260/A280suitable for downstream applications including qRT-PCR, RT-PCR, microarrays, NGS and more.
The kit purifies all sizes of RNA from large mRNA, lncRNA down to microRNA (miRNA) in the same fraction without the requirement of phenol. Isolate all RNA sequences at an equal rate irrespective of size. Moreover, when the RNA sequences are small (e.g. miRNA), the column binds small RNAs regardless of their GC content.
Isolate RNA after Purifying EVs and Exosomes
Ultracentrifugation, Exoquick, Filtration
| Cat. # | Name | Elution Volume | Plasma/Serum | Urine | Cell-Culture Media |
|---|---|---|---|---|---|
| 55000 | Plasma/Serum RNA Purification Mini Kit | 10 - 25 µL | 50 µL - 1 mL | 250 µL - 1 mL | 5 - 10 mL |
| 35300 | Total RNA Purification Micro Kit | 20 - 50 µL | 1 - 4 mL | 2 - 10 mL | 10 - 20 mL |
| 17200 | Total RNA Purification Kit | 50 - 100 µL | 4 - 10 mL | 11 - 30 mL | 20 - 35 mL |
Top 5 Reasons to Choose Silicon Carbide RNA Extraction Technology
About Norgen
The Company
Norgen Biotek was founded in 1998 by Dr. Yousef Haj-Ahmad. The company is located in a 24,000 square foot state-of-the-art facility in Thorold, Ontario, Canada a few blocks from Brock University. Norgen is an ISO 9001, ISO 13485 and Illumina Propel certified fully-integrated Canadian biotechnology company focusing on developing products for sample collection, sample preparation and sample detection, as well as providing comprehensive research services to the scientific community. Norgen’s ISO 9001 and ISO 13485 certifications indicate our commitment to manufacturing and selling high quality products, as well as our commitment to continually improving our company, our products and our quality management system. Our Illumina Propel certification emphasizes our proficiency with Illumina Next-Generation Sequencing (NGS) to support genetic and genomic research and provide high quality services to our customers.

Norgen's Technology
Norgen holds more than thirty issued and pending patents. Norgen has developed a unique platform technology based on a proprietary resin/matrix with many applications including the purification, concentration and clean-up of DNA, RNA, microRNA, proteins and exosomes from various specimen types. The efficient purification of these macromolecules is often the first step in diagnostics and in hundreds of molecular biological applications fueling discovery for genomics and proteomics. The novelty of this technology has been recognized by the National Research Council by awarding Norgen the prestigious "Innovation Leader Award for 2003".

Manufacturing Capability, Contract Manufacturing and OEM
Norgen develops and manufactures all of our products, including isolation and purification kits, sample collection and preservation devices, molecular diagnostic kits and NGS kits. We not only manufacture all the reagents and solutions for these kits, we also manufacture the spin columns associated with these kits. Norgen has a fully equipped manufacturing facility, including an ISO class 5 certified cleanroom used for high capacity liquid filling. Norgen also has tremendous expertise in OEM and contract manufacturing of kits and reagents for the molecular biology and diagnostics markets. This includes customized manufacturing of full purification/isolation kits, sample collection and preservation devices, as well as molecular biology reagents including buffers and RNA/DNA standards. Norgen’s contract manufacturing services are catered to each individual’s needs, and we are fully committed to supplying high quality products and services. We offer best-in-class products, fast delivery time, and personable, highly competent, and unparalleled services.

cGMP
All of Norgen’s products are manufactured according to the ISO 13485:2016 standard, which meets or exceeds cGMP compliance. From our fully validated equipment and manufacturing premises to our highly trained personnel, we meet all the requirements of GMP. All of our manufacturing processes are strictly controlled by SOPs, ensuring batch to batch consistency and full traceability for all products manufactured in our facility. In addition to the manufacture of own products and medical devices, we also offer contract manufacturing to the life science and medical device community.