- All-in-one solution for inclusion body protein isolation and purification
- Complete kit with Cell Lysis Reagent, Inclusion Body Solubilization Reagent, buffers and spin columns to purify proteins
- Isolate up to 12 mg of protein per spin column
- Fast and convenient spin column protocol
ProteoSpin™ Inclusion Body Protein Isolation Maxi Kit
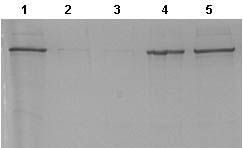
This kit provides everything required to isolate and purify inclusion body proteins from induced bacterial cultures. First a proprietary Cell Lysis Reagent is used to selectively lyse the cells and release inclusion bodies in their solid form. Next, inclusion bodies are dissolved and their contents released using the provided IB Solubilization Reagent. Inclusion body proteins are then further purified using spin columns for rapid and convenient buffer exchange and desalting.
The procedure is efficient and streamlined and can process up to 4 samples in approximately 2 hours. Each spin column is able to recover up to 12 mg of acidic or basic proteins from 100 mL of induced bacterial culture. Purified recombinant proteins are then ready for SDS-PAGE, 2D gels, Western blots, Mass Spectrometry analysis, and other applications.
About Inclusion Bodies
Bacteria are widely used for the expression of different proteins. However, 70-80% of the proteins expressed in bacteria by recombinant techniques are tyically contained in insoluble inclusion bodies (i.e., protein aggregates). The protein of interest found in these sub-cellular structures is often inactive, due to incorrect folding. The production rate of recombinant proteins stored in inclusion bodies is invariably higher than those synthesized as soluble proteins. The reason behind this is thought to be the resistance of insoluble proteins to proteolysis by cellular enzymes. In addition, separation of insoluble recombinant proteins in inclusion bodies is considerably easier than that of soluble proteins. These factors have been the major influences favoring scale-up of high-value proteins using bacterial fermentation for example. Procedures for the purification of the expressed proteins from inclusion bodies are often labour-intensive, time consuming and not cost effective. This kit provides the essential reagents for cell disruption, inclusion body solubilization and purification using spin column chromatography - all optimized to work together thereby simplifying the process and saving a tremendous amount of time and cost.
About Norgen
The Company
Norgen Biotek was founded in 1998 by Dr. Yousef Haj-Ahmad. The company is located in a 24,000 square foot state-of-the-art facility in Thorold, Ontario, Canada a few blocks from Brock University. Norgen is an ISO 9001, ISO 13485 and Illumina Propel certified fully-integrated Canadian biotechnology company focusing on developing products for sample collection, sample preparation and sample detection, as well as providing comprehensive research services to the scientific community. Norgen’s ISO 9001 and ISO 13485 certifications indicate our commitment to manufacturing and selling high quality products, as well as our commitment to continually improving our company, our products and our quality management system. Our Illumina Propel certification emphasizes our proficiency with Illumina Next-Generation Sequencing (NGS) to support genetic and genomic research and provide high quality services to our customers.

Norgen's Technology
Norgen holds more than thirty issued and pending patents. Norgen has developed a unique platform technology based on a proprietary resin/matrix with many applications including the purification, concentration and clean-up of DNA, RNA, microRNA, proteins and exosomes from various specimen types. The efficient purification of these macromolecules is often the first step in diagnostics and in hundreds of molecular biological applications fueling discovery for genomics and proteomics. The novelty of this technology has been recognized by the National Research Council by awarding Norgen the prestigious "Innovation Leader Award for 2003".

Manufacturing Capability, Contract Manufacturing and OEM
Norgen develops and manufactures all of our products, including isolation and purification kits, sample collection and preservation devices, molecular diagnostic kits and NGS kits. We not only manufacture all the reagents and solutions for these kits, we also manufacture the spin columns associated with these kits. Norgen has a fully equipped manufacturing facility, including an ISO class 5 certified cleanroom used for high capacity liquid filling. Norgen also has tremendous expertise in OEM and contract manufacturing of kits and reagents for the molecular biology and diagnostics markets. This includes customized manufacturing of full purification/isolation kits, sample collection and preservation devices, as well as molecular biology reagents including buffers and RNA/DNA standards. Norgen’s contract manufacturing services are catered to each individual’s needs, and we are fully committed to supplying high quality products and services. We offer best-in-class products, fast delivery time, and personable, highly competent, and unparalleled services.

cGMP
All of Norgen’s products are manufactured according to the ISO 13485:2016 standard, which meets or exceeds cGMP compliance. From our fully validated equipment and manufacturing premises to our highly trained personnel, we meet all the requirements of GMP. All of our manufacturing processes are strictly controlled by SOPs, ensuring batch to batch consistency and full traceability for all products manufactured in our facility. In addition to the manufacture of own products and medical devices, we also offer contract manufacturing to the life science and medical device community.